Addition cross-linking. Unsaturated polyester (UPE) resins. Vinyl ester (VE) resins.
PowerPoint
papers
Index

| Composites Design and Manufacture (Plymouth University teaching support materials) Addition cross-linking. Unsaturated polyester (UPE) resins. Vinyl ester (VE) resins. |
Lecture PowerPoint |
Review papers |
Subject Index |
 |
Further to the discussion of the condensation reaction in the context of both the polymerisation and the cross-linking (curing) of polymers, a second group of polymers exist in which the backbone chain is made by a condensation reaction with double bonds (unsaturation) in the backbone of the chain. Cross-linking is then achieved by an addition reaction through the opening of the double bonds at sites in two adjacent chains. The unsaturated polyester (UPE) laminating resins are generally viscous, yellowish materials of low (8-10) degree of polymerisation and molecular weight around 1000. The addition of a reactive diluent (usually styrene) eases working, often reduces cost and enhances the reactivity of the polyester. Styrene is a volatile organic compound (VOC) and may vaporise , but cross-linking occurs without the evolution of any by-products and UPE can hence be moulded without the use of pressure.
The molecular chains of the polyester can be represented as follows, where A is derived from the alcohol and B is derived from the unsaturated acid and hence contains the unreacted double bond (unsaturation):
| - | A | - | B | - | A | - | B | - | A | - | B | - |
With the addition of styrene [S], and in the presence of a catalyst and accelerator, the polymer chains can be cross-linked (cured) to form a three-dimensional network represented simplistically in 2-D as below. Note that diluting the resin with styrene will increase the number of styrene molecules in each crosslink and the cured resin will increasingly take on the brittle characteristics of polystyrene resin:
| - | A | - | B | - | A | - | B | - | A | - | B | - |
| | | | | | | ||||||||||
| S | S | S | ||||||||||
| | | | | | | ||||||||||
| - | A | - | B | - | A | - | B | - | A | - | B | - |
| | | | | | | ||||||||||
| S | S | S | ||||||||||
| | | | | | | ||||||||||
| - | A | - | B | - | A | - | B | - | A | - | B | - |
Figure 1: Simplified representation of unsaturated polyester resin (AB) with styrene (S) cross links:
in practice the
network will be three-dimensional.
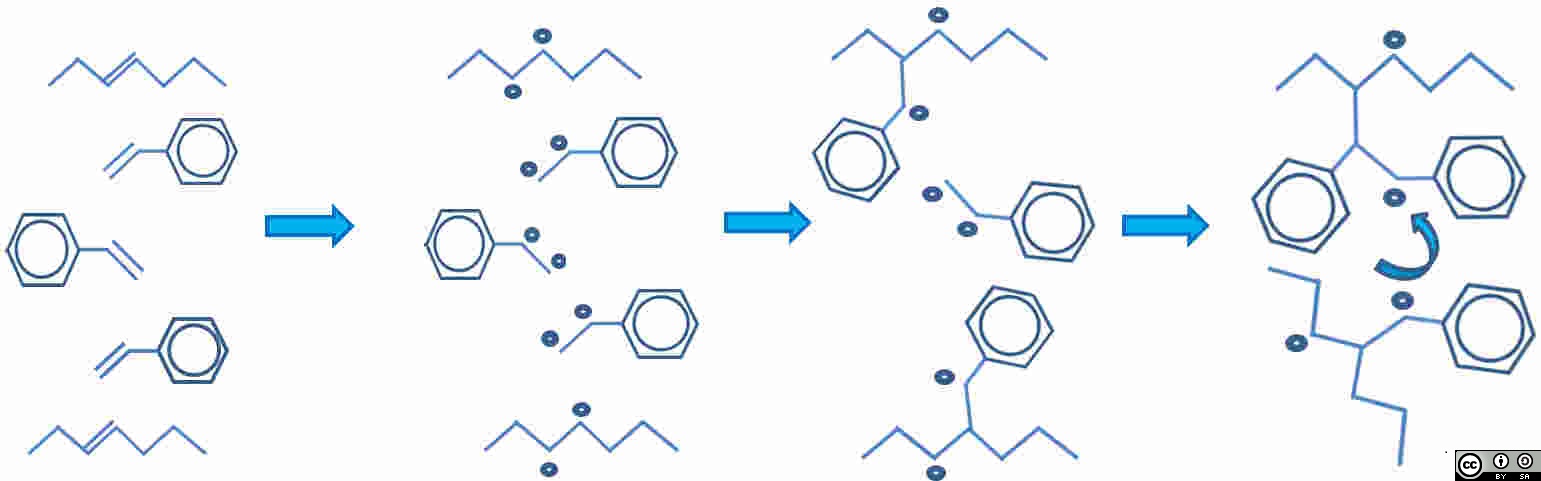
| Unsaturated polyester and styrene both have double bonds. Polyester chain top and bottom, with 3 styrene molecules between. |
A broken bond = two free radicals. Assume all double bonds break. Five double bonds are now ten free radicals |
Free radicals pair-up to make new bonds |
Molecules move closer together and rotate to form cross-link. Net shrinkage of system results. Note two remaining reactive sites. |
Figure 2: The progress of the addition curing reaction in unsaturated polyester.
A curing system must be blended into the resin before application to the fibres. Room temperature cures are available for the manufacture of large structures, while press mouldings may be cured at elevated temperatures (about 100°C). Peroxides, including acetylacetone peroxide (AAP), cyclohexanone peroxide (CHP), or methyl ethyl ketone peroxide (MEKP) are common catalysts. They are used with cobalt (or vanadium) naphthenates as accelerators. The peroxide and the accelerator should never be brought into direct contact as they form an EXPLOSIVE mixture. Usually the accelerator is well dispersed into the resin before the peroxide is added. Gel times can be varied from five minutes to several hours. The peak exotherm at gelation may reach 200°C, particularly in bulky components.
Benzoyl peroxide [BPO: 94-36-0] can be used for room temperature curing of unsaturated polyester resins in the presence of certain aromatic tertiary amines. BPO is not accelerated by the naphthenates.
Further reading:
Vinyl Ester (VE) resins
Vinylester resins are normally an epoxy resin backbone grafted with an unsaturated carboxylic acid to permit cure by an addition reaction. They generally possess the good tensile strength and chemical resistance of the epoxy resin, but with the faster curing times and high volumetric shrinkage on cure associated with the unsaturated polyesters..
Bismaleimides
The bismaleimide [BMI] resins are cured principally by a network reaction but this may be supplemented by a radical (addition) reaction.